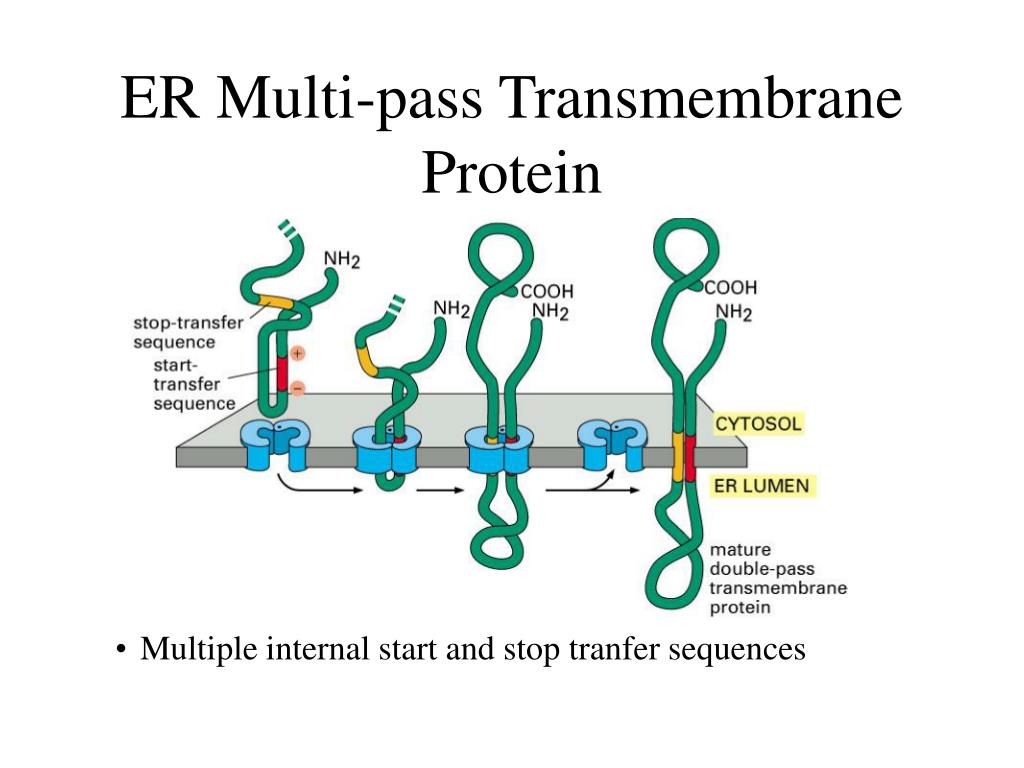 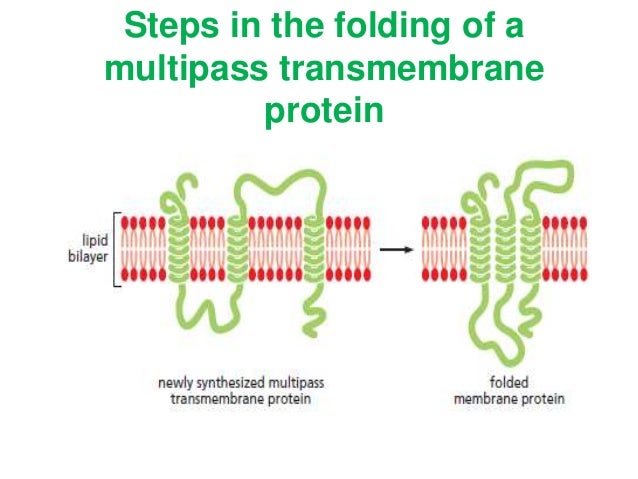
Some membrane proteins do not extend into the hydrophobic interior of the lipid bilayer at all they are instead bound to either face of the membrane by noncovalent interactions with other membrane proteins (examples 7 and 8 in Figure 10-17). This enzyme cuts these proteins free from their anchors, thereby releasing them from the membrane. Proteins bound to the plasma membrane by a GPI anchor can be readily distinguished by the use of an enzyme called phosphatidylinositol-specific phospholipase C. While still in the ER, the transmembrane segment of the protein is cleaved off and a glycosylphosphatidylinositol (GPI) anchor is added, leaving the protein bound to the noncytosolic surface of the membrane solely by this anchor (discussed in Chapter 12). The proteins in example 6, however, are made as single-pass transmembrane proteins in the ER. The lipid-linked proteins in example 5 in Figure 10-17 are made as soluble proteins in the cytosol and are subsequently directed to the membrane by the covalent attachment of a lipid group (see Figure 10-18). (A) A fatty acid chain (myristic acid) is (more.) The covalent attachment of either type of lipid can help localize a water-soluble protein to a membrane after its synthesis in the cytosol. Membrane protein attachment by a fatty acid chain or a prenyl group. Thus, the surface that the cell presents to the exterior is rich in carbohydrate, which forms a cell coat, as we discuss later. Like membrane lipids, membrane proteins often have oligosaccharide chains attached to them that face the cell exterior. A typical plasma membrane is somewhere in between, with protein accounting for about 50% of its mass.īecause lipid molecules are small compared with protein molecules, there are always many more lipid molecules than protein molecules in membranes-about 50 lipid molecules for each protein molecule in a membrane that is 50% protein by mass. By contrast, in the membranes involved in ATP production (such as the internal membranes of mitochondria and chloroplasts), approximately 75% is protein. In the myelin membrane, which serves mainly as electrical insulation for nerve cell axons, less than 25% of the membrane mass is protein. Accordingly, the amounts and types of proteins in a membrane are highly variable. It is the proteins, therefore, that give each type of membrane in the cell its characteristic functional properties. Although the basic structure of biological membranes is provided by the lipid bilayer, membrane proteins perform most of the specific functions of membranes.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
